|
Chemistry 350 Laboratory - Fall Semester 2012 – Professor T. Nalli, Winona State University Expt #5. Dehydrohalogenation of
2-Bromoheptane
revised on 12/13/2012 Relevant textbook readings – Mohrig,
Chapter 11, 12. Klein, Chapter 8
Overview
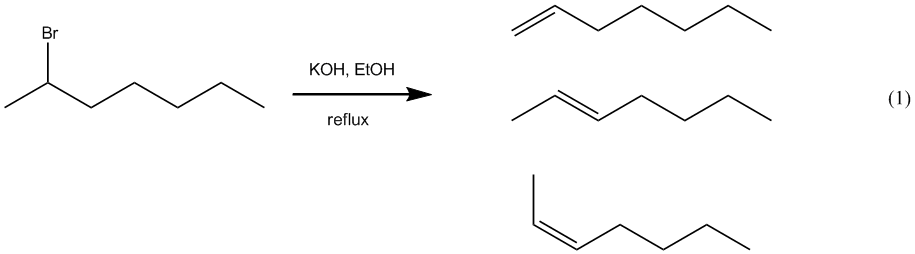
– In this experiment, we will react 2-bromoheptane with the strong base, potassium
hydroxide, to obtain an alkene product (eq 1). The
expected mechanism for this reaction is E2 and three different alkenes are
possible. The main goals of this experiment are to determine which alkene is actually
formed as the major product and to determine amounts of any minor products that are also formed.
Procedures Safety Wear gloves when measuring out the
reactants and throughout the extraction procedures. Ethanolic
KOH is very caustic and skin contact should be avoided. If you do get KOH on
your skin (you will not immediately feel a burning sensation but you can tell
it’s there by a slippery/soapy sensation on your skin) make sure to
immediately rinse with cold water for at least five minutes. Running the reaction Add 0.5 g KOH, 1.0 mL
95% ethanol, and a stir bar to a 5-mL round bottom flask (rbf). Stir or swirl briefly then attach a condenser and reflux for 5 min before adding 0.65 mL 2-bromoheptane by pipet to the rb flask. Reflux the solution for 60 min.
Work-up Cool the solution to
near room temperature and then transfer it into a reacion tube containing 1.0 mL
of cold water. Cap the tube and carefully mix and shake the contents being
careful to vent frequently. Let the tube stand for 5 min.
For the procedures in the following paragraph, make sure
to identify the organic and aqueous layers correctly! See section 11.2 –
“Practical Advice on Extractions” in Mohrig. You
can also test separated aqueous layers by adding a few drops of water to them
to make sure that the water dissolves in. If the presumed aqueous layer is
actually organic then the added water drops will not dissolve. Use a pipet to
transfer the organic layer to another test tube. Then wash the organic layer
with 1.0 mL H2O being careful to vent as needed. Allow the layers to
separate and pipet out the water layer into a waste beaker to be discarded
later. (As a general rule, never discard any material from a reaction until
the final purified product is obtained and verified). Wash the organic layer
with 1.0 mL H2O twice more, each time transferring the aqueous layer
into the waste beaker.
Dry the organic layer
over sodium sulfate. Use a Pasteur pipet to remove the liquid from the drying
agent and transfer it into a dry, pre-weighed vial.
Weigh the vial with the product to
determine the yield.
Characterization
of Product
Obtain a proton NMR spectrum of the product. Prepare a sample for GC-MS analysis by adding 1 uL of the product to approx 10 mL pentane in a clean vial. One group will also be asked to obtain a C-13 NMR of their product. Report Literature Spectra - Do not include literature spectra data in this report. Instead GC-MS and NMR spectra of each of the possible alkene products and the 2-bromoheptane starting material were obtained here at WSU and are being provided. (See additional details below.) Proton NMR. Proton NMR data files for 1-heptene, E-2-heptene, and Z-2-heptene are available in the class storage folder. Process each of these spectra and summarize the data for all four compounds in a single results table (Table 1). Be aware that complex splitting patterns are expected for the Hs of the double bonds, which are expected to resonate in the vicinity of 5-6 ppm.. This is due to the (n + 1)(m +1) rule applying here because of the fact that the Hs causing splitting are not equivalent and have quite different coupling constants (J values). (see Klein p 745 and Mohrig p 346). Use the 1H NMR data on the pure compounds to assign peaks in the 1H NMR of the product mixture and include these findings in Table 1.
C-13 NMR. 13C-NMR data files for the three alkenes are also available in class storage. Process each of these spectra and summarize the data for the three compounds in a single results table (Table 2). Use the13C NMR spectrum obtained by the selected team as indicative of what a 13C NMR spectrum of your product would have looked like. Assign peaks in the above 13C NMR spectrum based on the spectra of the pure compounds and include these findings in Table 2.
GC-MS. You will be provided with separate printouts of GC-MS results for all three alkenes and the starting material alkyl bromide. Summarize this data, including the retention time of the compound as well as the m/z values of the M+ and base peaks plus a few other strong or significant MS peaks in a table (Table 3). Use the retention times of the pure compounds to assign the peaks in the GC chromatogram (top graph) of the product mixture and include these findings in Table 2.
Bonus: An unexpected product shows up at 7.0 min in the GC-MS. Use the mass spectrum of this product to determine its identity and propose a mechanism for how it forms. Reexamine the NMR spectra to check for peaks due to this side product. Label these peaks and include the data in tables 1, 2, and 3. Summary Table. Use Table 4 as a master table to summarize all of the most important findings of the experiment. Include the absolute yield (g), theoretical yield (g) and percent yield. Also include what the NMR, GC-MS, and IR spectra reveal about the product mixture, especially as pertains to the relative amounts of the three alkenes formed. |