Expt #4. Preparation of 2-Naphthyl Butyl
Ether
Relevant textbook readings
– Mohrig, Chapter 6-7, 10, 15. Klein, Chapter 7
References
- (1) Experiment adapted from Esteb, J. J.; Magers,
J. R.; McNulty, L.; Morgan, P.; Wilson, A. M J.
Chem. Educ.2009, 86,
850. (2) Also see Wenkert, E.; Youssefyeh, R.
D.; Lewis, R. G. J. Am. Chem. Soc. 1960,
82, 4675.
Overview – The SN2 reaction is a
versatile and stereospecific reaction that can be
used for the preparation of a multitude of
functional group families. These facts contribute to
its great importance as a method for organic
synthesis. The reaction generally involves a
nucleophile attacking the electrophilic carbon of an
unhindered alkyl halide (or alkyl sulfonate)
displacing the halide (or sulfonate) leaving group.
In the reaction that you will
be carrying out, the nucleophile is the naphthoxide
ion formed from the reaction of 2-naphthol
with sodium hydroxide. The alkyl halide is
1-iodobutane and the expected product is butyl
naphthyl ether (eq 1).
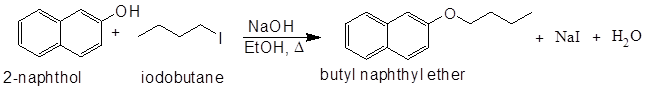 (1)
(1)
Procedures
Running the reaction.
To a
100-mL round bottom flask (rbf), add 0.56 g of
NaOH and 1.0 g of 2-naphthol. Add 20 mL ethanol
and a few boiling chips to the flask. Reflux the
solution for 15 minutes or until all of the solids
have dissolved. After 15 minutes add 1.0 mL
1-iodobutane by Pasteur pipet through the top of
the condenser and then reflux for 1 h.
Work-up procedures.
Cool the reaction mixture on
a cold water bath for 5 min. Slowly pour the
solution over 25 g ice in a 250-mL beaker. Rinse
the rbf with 25 mL ice-cold water and add this to
the beaker. Swirl or stir the contents of the
beaker until all of the ice has melted. Make sure
the mixture is thoroughly chilled on an ice bath
and then collect the solid product from the ice
cold solution by vacuum filtration. Wash the
product with a few mL of ice-cold water and then
dry it somewhat by allowing air to flow through
the solid for 5-10 min.
Characterization
of Product
Obtain the melting point. (A
literature value can be found in the references).
Please note that your product is not expected to
extremely pure because it was not subjected to a
final recrystallization step.
Obtain the 1H NMR
spectrum using CDCl3 as the solvent.
Obtain the IR spectrum.
Solids can be difficult to obtain an IR spectrum
for so we will take advantage of the very low mp
of this compound and melt it to a liquid before
obtaining the IR. Place about 25 mg of the solid
product on a salt plate and then place the salt
plate on a warm surface (the top of the GC-MS
ionization chamber works well for this). Once it
is melted, add a second salt plate and obtain the
spectrum.
Prepare a sample for GC-MS
analysis. Dilute a few drops of your NMR sample in
10 mL DCM in a clean labeled vial.
Report
Please try to integrate the
answers to the assigned questions into your results
and conclusions section for this report (rather than
answering each sequentially).
1H
NMR - Be careful to first identify and
label solvent peaks including CHCl3 and
water (usually at 1.55 ppm in CDCl3
solvent.) Integrate all peaks and normalize so that
the integrations directly show the number of
hydrogens for each resonance. Several protons'
resonances overlap to form messy multiplets. These
multiplets can still be identified as to which
protons are included and should be reported in the
results table as multiplicity = "m" and with the
range of ppm values that they span given as the
chemical shift.
Questions:
- Ethanol is not usually a
solvent used for SN2 reactions.
Why not? Can you predict an undesirable side
reaction that might occur using the ethanol as a
solvent that would not occur if we used THF as a
solvent?
- Why is it important to react
the naphthol with sodium hydroxide first before
adding the electrophile? What unwanted side
reaction would be more likely to occur if all
three reactants were added together at the start
and the mixture refluxed?
- Why is it important to fully
cool the product in an ice bath prior to vacuum
filtration? Why do is the product washed
with ice water instead of water straight from the
tap?
|