Expt #1. Synthesis of Acetaminophen
Relevant textbook readings – Mohrig, Chapter
3-7, 10, 14, 15.
References - (1)
Experiment adapted from Synthesis of Acetaminophen
(Experiment). (2020, July 25). Retrieved June 23,
2021, from https://chem.libretexts.org/@go/page/61312accessed 6/23/2021.
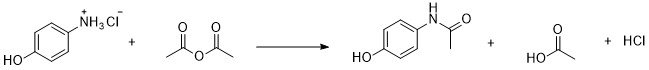
Overview – In this lab you will react
p-aminophenol hydrochloride with acetic anhydride to
form the common pain reliever, acetaminophen as the
main product. Acetic acid is also formed in this
reaction as a side product. The obtained acetaminophen
will be purified by recrystallization and then
characterized through a melting point (mp)
determination.
The goals of the lab are to (1) obtain a good yield
of pure acetaminophen (2) become familiar with some of
the glassware and equipment commonly used in the
organic chemistry lab (3) learn how to determine the
mp of a solid and how to interpret the result and (4)
learn how to purify a solid through the process of
recrystallization. The lab will also provide an
opportunity to learn how to take good lab notes and
write an effective lab report.
Procedures
Running the Reaction
-
Place 0.56 g p-aminophenol
hydrochloride solution in a 25-mL round bottom
flask (rbf).
-
Clamp up the rbf and add 7 mL water
and a magnetic stirbar.
-
Begin stirring and attach a reflux
condenser and heat the mixture on a sand bath to
a gentle reflux (fig 7.1a, p 60).
-
While waiting for the mixture to reach
reflux prepare a buffer solution of 0.5 g
sodium acetate trihydrate in 1.5 mL water.
- Use a Pasteur pipet to add the buffer solution
to the stirring solution in the rbf. Do the
addition through the top of the reflux condenser.
-
Immediately add 0.4 mL of acetic
anhydride (also through the top of the reflux
condenser).
-
Continue refluxing for 10 min.
Work-up
- Cool the solution on an ice bath, stirring and
scratching with a glass rod until the crude
acetaminophen begins to crystallize. After
crystallization begins, allow the solution to sit
on the ice bath until crystal growth appears to
have reached completion.
- Collect the product by vacuum filtration on a
Buchner funnel. (fig 10.6, p 110) Wash the
crystals once with 1-2 mL of ice cold water and
continue pulling air through them for 5-10 min so
that they are reasonably dry.
- Transfer the crude product to a watch glass or
weighing boat and determine the yield. Calculate
the percent yield and record it in your lab notes.
- Recrystallize all but 100 mg of the crude
acetaminophen from water. (Use the miniscale
procedure described in chap 15.4-15.5).
- Record the weight of the recrystallized
acetaminophen.
- Calculate the % recovery from the
recrystallization (mass xtals/mass crude x 100) as
well as the final % yield.
Characterization
of Product
Obtain the melting points of both the final
crystals and the
crude as well as of a commercial sample of
acetaminophen.(Chapter 14)
|
