Dr. Nalli's Chemistry 351 Laboratory - Spring Semester 2010
Expt #4. Diels-Alder Reaction of alpha-Phellandrene with Maleic Anhydride
Relevant textbook readings - Smith - chapter 16.12-16.14. Pavia - Technique 11, pp 189-210. Mohrig Technique 9, pp 100-115.
Prelab - Make sure you calculate the volume of phellandrene and the mass of maleic anhydride to use and include both in your table of reactants and products. Also make sure to include the theoretical yield of the product. As an additional assignment for this pre-lab, a prediction of the 13C NMR spectrum of the product is required. List the peaks expected along with their approximate chemical shifts.
Overview - You will carry out the Diels-Alder reaction of the terpene natural product, alpha-Phellandrene, with Maleic Anhydride. The resulting product could conceivably be any of four possible diastereomers as shown in the equation below.
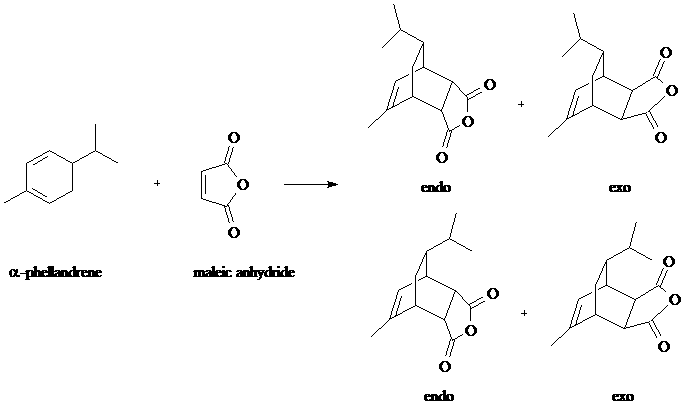
We can predict that the reaction will be stereoselective, forming predominantly the stereoisomer at top left. We dismiss the bottom two structures based on prohibitive steric hindrance from the isopropyl group and, based on the endo rule, we expect an endo rather than exo product. The endo rule is explained by bonding interactions in the transition state between pi bond substituents on the dienophile (in this case the C=O groups) and the p orbitals at carbons 2 and 3 of the diene.
We will run the reaction and obtain a crystalline product that will be thoroughly characterized by 1H, 13C, DEPT, and HMQC NMR. An IR spectrum and mp will also be obtained (lit mp 126-127).
Procedure
Reaction:
- In a 25-mL round-bottom flask put 0.010 mole of maleic anhydride and 0.010 mole of alpha-Phellandrene.
- Add 5 mL of diethyl ether, attach a reflux condenser, and heat on a hot water wath for 60 min.
- Allow to cool slowly to room temperature then cool on an ice bath to maximize crystal formation.
- Collect the crystals by vacuum filtration.
Purification:
- Recrystallize the product from ethyl acetate. (See technique 9 in Mohrig for all that this entails!)
- Get the crystals as dry as possible by sucking air through on the Buchner funnel for approx 15 minutes or by air drying for one week.
Analysis:
- Obtain the IR spectrum using the cast-film method of sample preparation. (see p 239 in Mohrig).
- Prepare a sample for NMR spectroscopy and obtain a proton NMR and one of the following: 13C NMR, DEPT-135, HMQC, HMBC, or COSY. The instructor will assist.
Modeling:
- Build a model of the expected product using HyperChem. Optimize it using a semi-empirical MO calculation.
- Print out an image of the HyperChem model and include it with your report.